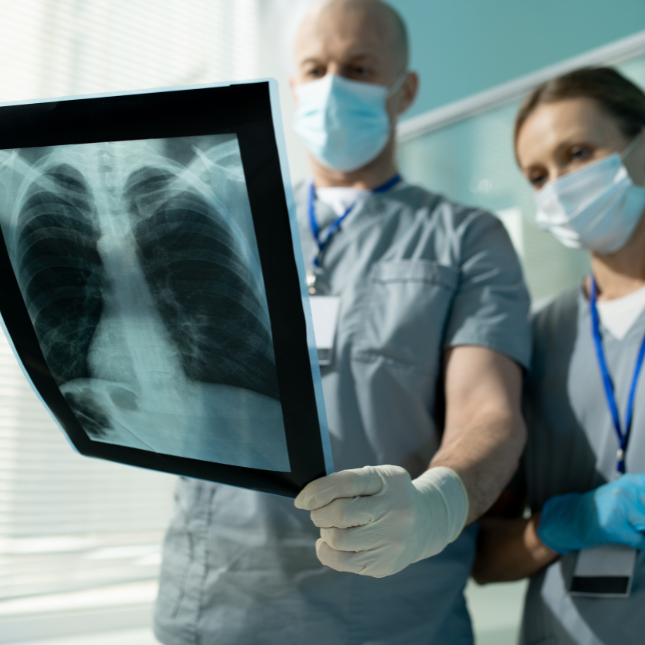
Combatting multi-drug resistant TB in Central Asia and worldwide requires strengthening manufacturing, laboratory and regulatory systems, and pharmacovigilance.
We work to ensure the quality and availability of tuberculosis medicines across the supply chain to limit the spread of multidrug-resistant tuberculosis (MDR-TB). This includes implementing innovative, expedited pathways to register quality-assured TB medicines in Central Asia, where MDR-TB is a persistent threat. It also means strengthening diagnostic and medicine quality control laboratories to better diagnose patients and ensure the quality of TB medicines.
In addition, we work with manufacturers to improve their compliance with global standards and best practices, including developing staff capacity and technical know-how, as well as preparing TB product dossiers for submission and review. Once TB products are approved for market use, we also work with national partners to monitor product safety to help detect any serious potential side effects, or adverse events, that might compromise TB treatment.